Shake Test
What is "Shake test”?
Shake test is used for determining if an adsorbed vaccine has been frozen anytime in the past. The test is valid for "adsorbed vaccines” only, such as DPT, DT, TT, Pentavalent vaccine and Hepatitis-B.
What is "Shake test”?
Shake test is used for determining if an adsorbed vaccine has been frozen anytime in the past. The test is valid for "adsorbed vaccines” only, such as DPT, DT, TT, Pentavalent vaccine and Hepatitis-B.
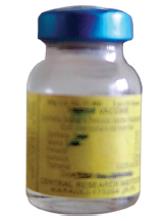
Diphtheria, Pertussis, and Tetanus(DPT; Triple Antigen)
Identification features
The vaccine is available in small glass bottles mostly having a blue metallic cover.
Type of vaccine
Killed (diphtheria toxoid, tetanus toxoid, and killed bacteria of pertussis)
Site of injection
Anterolateral aspect of LEFT mid-thigh
Type of Vaccine
Inactivated polio virus (killed virus)
COMPOSITION
Inactivated poliovirus type 1 - 40 D antigen units
Inactivated poliovirus type 2 - 8 D antigen units
Inactivated poliovirus type 3 - 32 D antigen units
Number of doses per vial
Vaccine comes in: 1-dose, 5-dose, and 10-dose vials
The Government of India is acquiring 5-dose and 10-dose vials currently
Type of vaccine vial monitor (VVM) on the vial
Type 7
Type of Vaccine
Live attenuated liquid vaccine
COMPOSITION
Monovalent vaccine containing live attenuated rotavirus derived from a neonatal strain isolated in India
Number of doses per vial
Rotavirus vaccine supplied under UIP has 10 doses per vial.
Type of Vaccine
Killed
Liquid form (liquid pentavalent vaccine) is available under UIP.
COMPOSITION
Each dose of 0.5 ml contains
1. Diphtheria Toxoid
2. Tetanus Toxoid
3. B. pertussis (whole cell)
4. HBsAg (rDNA)
5. Purified capsular Hib Polysaccharide (PRP)
Number of doses per vial
The liquid pentavalent vaccine (LPV) in the UIP (Universal Immunization Program, India) is available as a multi-dose vial with 10 doses per vial.
Identification features of BCG vaccine (Fig. 4.1)
• The vaccine is available in dark bottles to protect from sunlight.
• Some bottles have a long neck as the vaccine is packed in vacuum.
Type of vaccine
• Live
• Freeze dried powder
Dose
For institutional deliveries: 0.05 mL at the time of birth or up to age of 4 weeks
• In case of home delivery, if given at 6 weeks: 0.1 mL (along with Pentavalent – 1 and other routine vaccines)
Advocacy is a process for raising awareness, especially among decision-makers and service providers, to ensure that pentavalent vaccination is available for all targeted children.
For pregnant women
Early in pregnancy
Td-1* 0.5 mL Intramuscular Upper arm
4 weeks after TT-1
Td-2 0.5 mL Intramuscular Upper arm
*If the pregnant woman has received two Td doses in a pregnancy within last 3 years, only one Td-booster is required during the present pregnancy
For infants
Yellow non – chlorinated bag
Which categories waste goes into this?
1. Human anatomical waste
2. Animal anatomical waste
3. Soiled waste
4. Expired medicine
5. Chemical waste
6. Discarded linen or bedding
7. Microbiological waste
The term, ‘incinerate’ means, to burn something until nothing is left but sterile ash.
In an incinerator, the high levels of heat are kept inside the furnace or unit so that the waste is burnt quickly and efficiently.
‘Flue gases’ are generated from this waste burning process.
In a double chamber incinerator, these flue gases are also incinerated in the second chamber before being sent to the air pollution control device
The flue gases are cleaned of pollutants before they are dispersed in the atmosphere.
Ash after incineration is deep buried