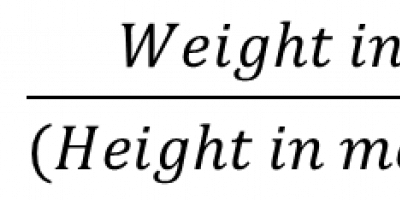You are here
Blogs
Confidence Interval – A Beginner’s Guide
If we want to measure a parameter of a population e.g. heights of the men in a city having 1,70,000 men:
One way can be that we measure the heights of ALL the 1, 70,000 men and calculate the mean.
This way we are sure we know the true mean of the population’s height (Population mean).
This may be a mammoth exercise and one may be hesitant in doing this.
So, mostly one chooses a sample from the study population, measures the heights of the participants and calculates the mean height of the sample.
विटामिन A की कमी से बिटोट स्पॉट का गठन क्यों होता है?
विटामिन ए की कमी से आंख की नमी एवं चिकनाई कम हो जाती है
इस से कंजाक्तिवा की कोशिकाएं शुष्क हो जाती हैं।
यह शुष्क और केरटिन्युक्त कोशिकाएं आँख में झड़ जाती हैं। यह अब एक मलबा होता है।
प्रत्येक पलक झपकने के साथ, यह मलबा आंख के केंद्र की ओर स्वाइप होता है।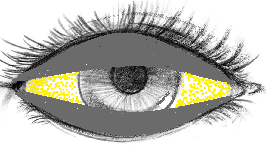
Why do Bitot’s Spots form in Vitamin A deficiency?
Vitamin A deficiency reduces mucus lubrication of the eye
•Which leads to drying of the conjunctiva and keratinization of its epithelial cells
These dry and keratinized cells are desquamated into the conjuctival sac as debris
With each blink, the eyelids swipes the debris toward the center of the eye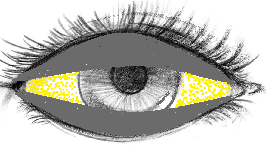
Null-Hypothesis: Epidemiology – a Beginner’s Guide
Research begins with ‘Null Hypothesis’ as the baseline.
Data is then gathered and analyzed to determine if there is enough evidence to reject the Null hypothesis in favor of an alternative hypothesis.
The evidence to reject or accept the Null hypothesis lies in calculating the p-value.
P-Value: Epidemiology – a Beginner’s Guide
In epidemiology, ‘p-value’ stands for ‘Probability – Value’
Value of probability or p-value ranges from 0 (zero) to 1 (one)
• A p-value of 0 means there is no chance of the event happening e.g. getting a seven on a dice
• A p-value of 1 means a 100% chance of the event happening e.g. Sun will rise in the east and set in the west.
Probability of WHAT?
Probability of the ‘Null – Hypothesis’ being true
स्वस्थ्य जीवनशैली सम्बंधित कुछ सलाह
खाने पीने का अनुशासन
• एक बार में कम खाना लें (छोटे बर्तन का प्रयोग करें) ।
• यदि आवश्यक (व्रत इत्यादि ) न हो तो, नाश्ता, दोपहर एवं रात्रि भोजन ना छोड़ें ।
• छोटे-छोटे कौर लें और प्रत्येक कौर का आनंद लेते हुए धीरे धीरे खाएं ।
• पूरे दिन के भोजन की डायरी बनायें । इसमें खाये गयी हर चीज़ की ईमानदारी से एंट्री करें ।
• पेट भरने का एहसास होते ही खाना बंद कर दें , पूरी प्लेट ख़त्म करने पर ज़ोर न दें ।
• खाद्य पैकेट के लेबल पर कैलोरी और अन्य पोषक तत्वों की जाँच करें ।
मोटापे के बारे में दिलचस्प तथ्य: वजन घटाने की यात्रा से पहले जानें
मोटापा कब कहा जाता है? जब बी एम् आई (BMI) का स्तर जरूरत से ज़्यादा होता है। जिस स्तर के ऊपर मोटापे के दुष्प्रभाव शरीर पर पड़ने लगे।
1. यूरोपीय मूल के लोगों की तुलना में, भारतीय मूल की लोगो को मोटापे से होने वाले स्वास्थय सम्बन्धी खतरे कम बीएमआई (बॉडी मास इंडेक्स) के स्तर पे ही दिखने लगते हैं।
इसलिए, मोटापे के लिए बीएमआई की सीमा उनके लिए कम रखी गयी है।
• ओवरवेट (ज्यादा वजन) के लिए BMI का कटऑफ: BMI ≥23 kg/m2 लेकिन <25 kg/m2 (पुरुषों और महिलाओं दोनों के लिए)
Some Lifestyle advice
Eating Habits
Take smaller portions at a time (consider using smaller container)
Do not skip major meals
Avoid snacks between meals
Take small bites and savor each bite
Maintain daily food intake record
Stop eating immediately when you feel full. Do not insist on finishing off the plate’s contents.
Check the label of the food packet for calories and other nutrients content
Smart Facts about Obesity – Must Know before Beginning the Weight Loss Journey
Overweight and obesity are defined at lower levels of BMI (Body Mass Index) for Indian population. This is because the health risks of obesity occur at a lower level of BMI in Asian Indians as compared to Caucasians.
Overweight: BMI ≥23 kg/m2 but <25 kg/m2 (irrespective of gender)
Generalized obesity (GO): BMI ≥ 25 kg/m2 (irrespective of gender)
Abdominal obesity (AO): Waist circumference (WC) ≥ 90 cm for men and ≥ 80 cm for women
How to measure your waist circumference:
• Feel and locate the lowest point of your ribs